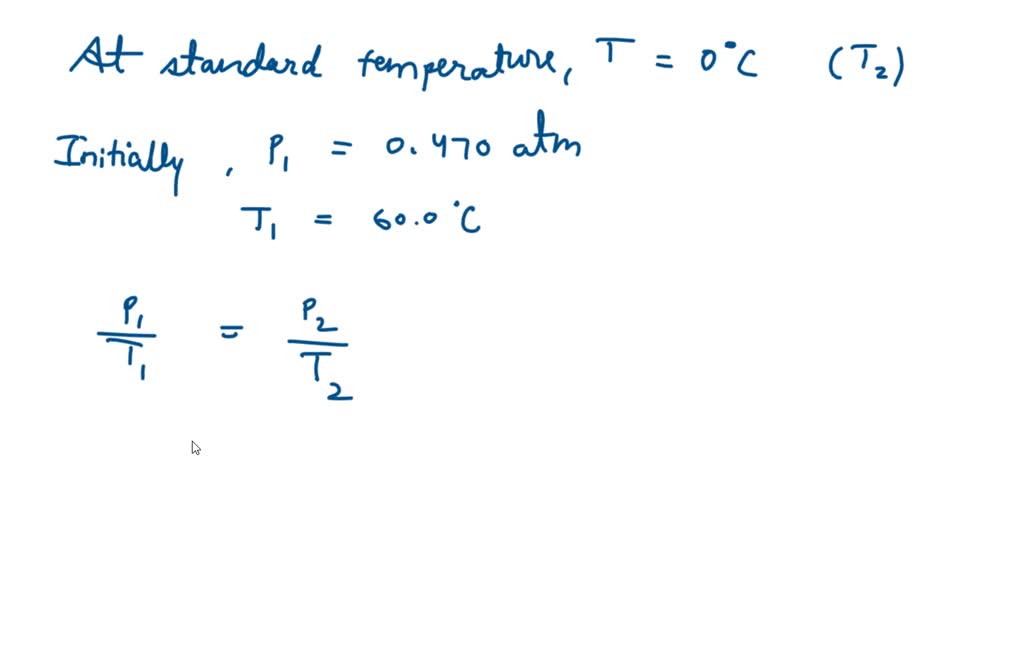At 546^oC and 1 atm pressure, 0.062 grams of gas occupies a volume of 33.6 ml . The molecular weight of the gas is:

SOLVED: Part A The pressure inside hydrogen-filled container was 2.10 atm at 21 C. What would the pressure be if the container was heated to 95 "C ? Express your answer with

Gas Laws. A. Characteristics of Gases Gases expand to fill any container. –random motion, no attraction Gases are fluids (like liquids). –no attraction. - ppt download
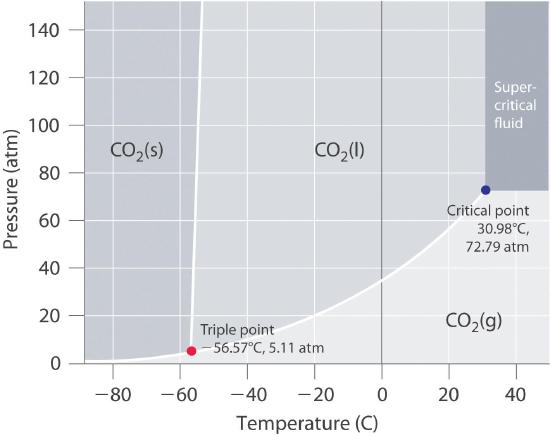
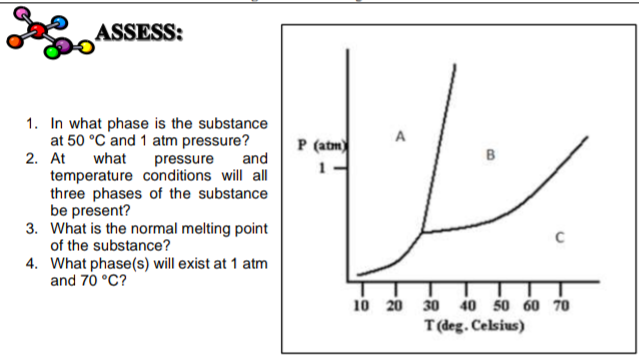
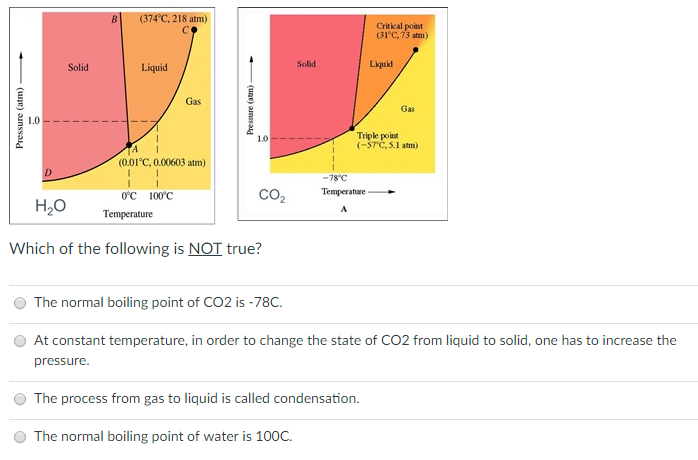
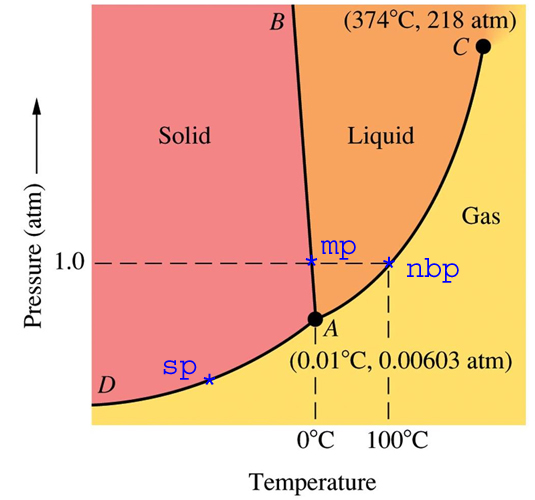
Using the phase diagram for CO_2, what phase is carbon dioxide in at -20 C and 1 atm pressure? | Socratic

A sample of a gas at 100^(@)C and 0.80 atm pressure has a density of 1.15 g L^(-1). What is the molecular weight of the gas?

SOLVED:Helium gas in a 2.00-L cylinder is under 1.12 atm pressure. At 36.5°C, that same gas sample has a pressure of 2.56 atm. What was the initial temperature of the gas in