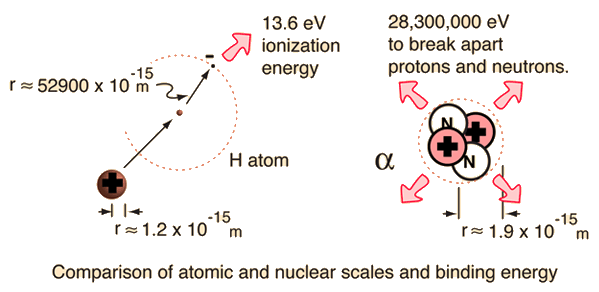
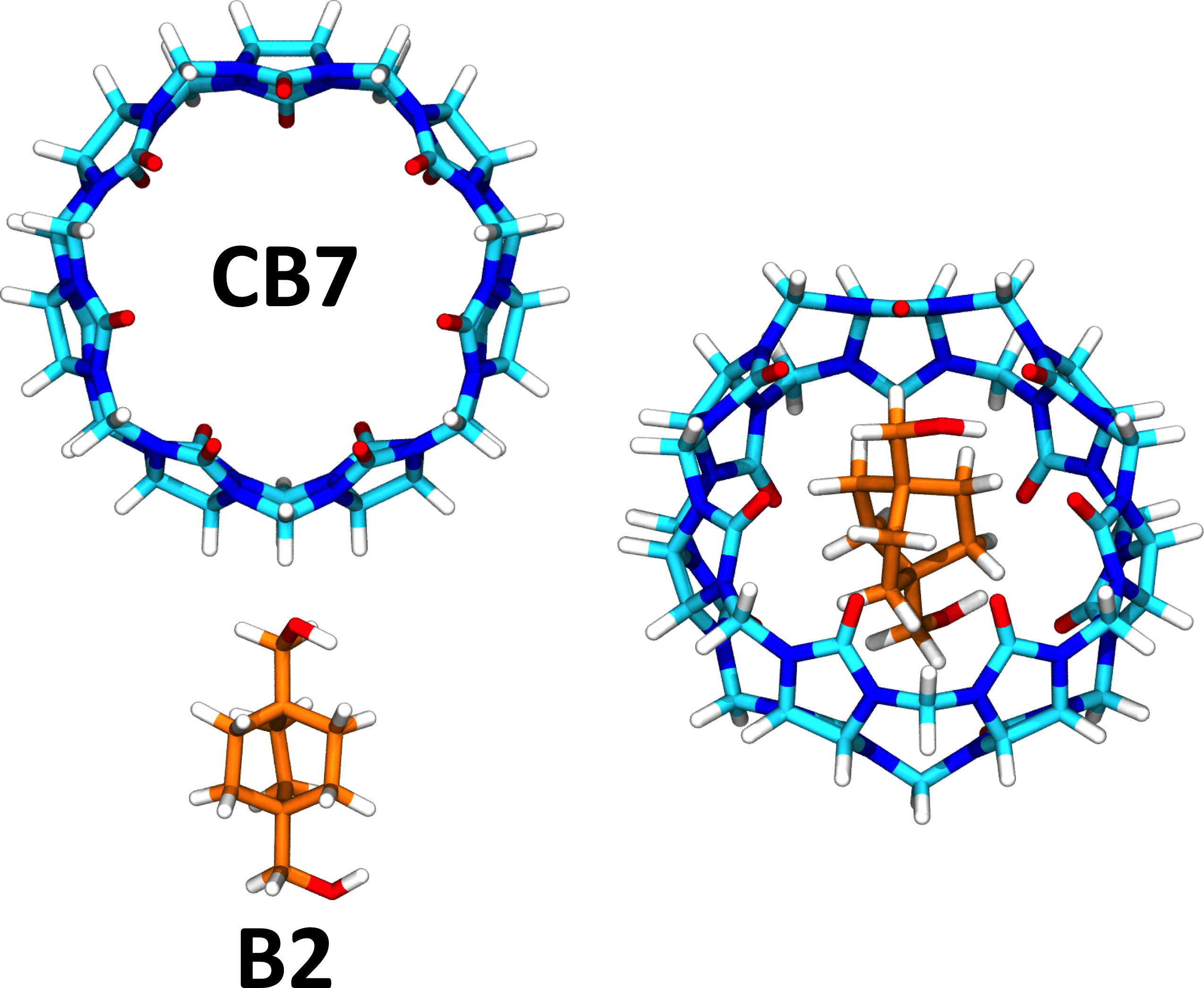
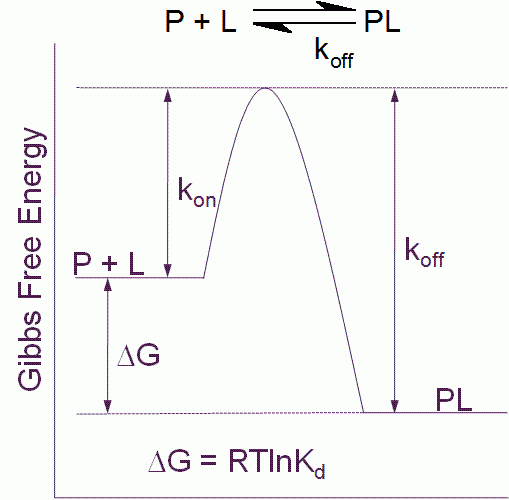
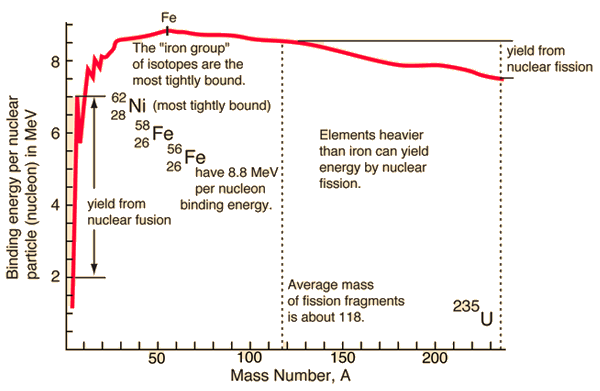
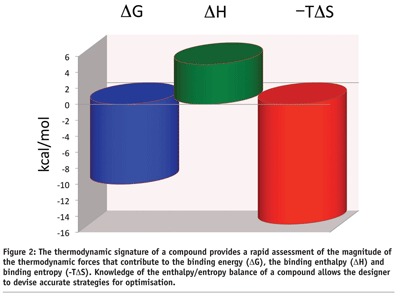
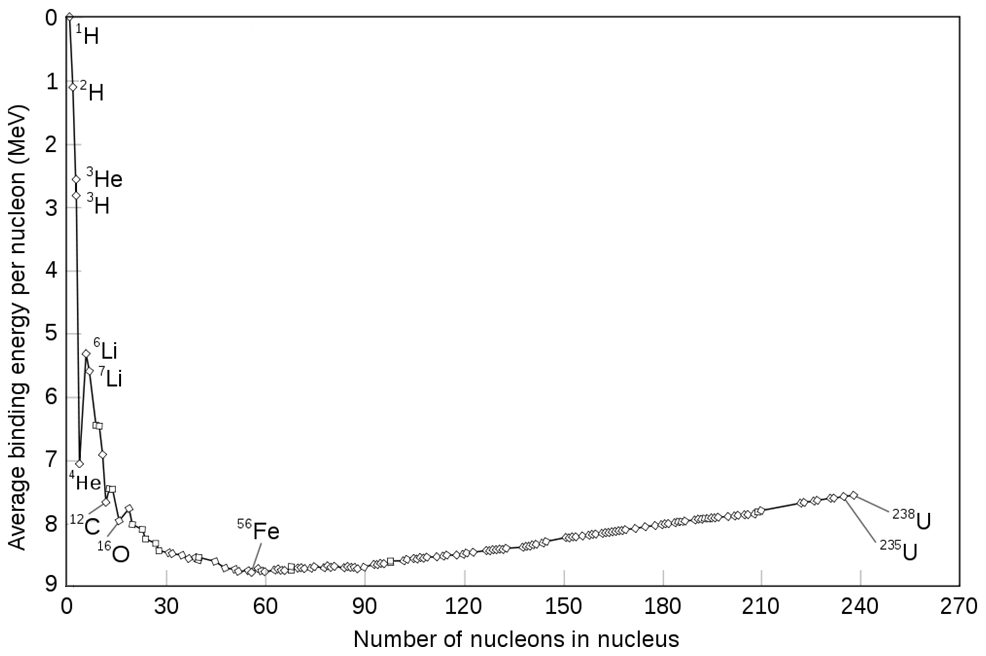
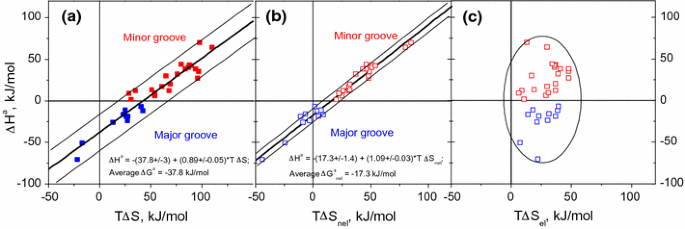
Ligand binding free energy and kinetics calculation in 2020 - Limongelli - 2020 - WIREs Computational Molecular Science - Wiley Online Library
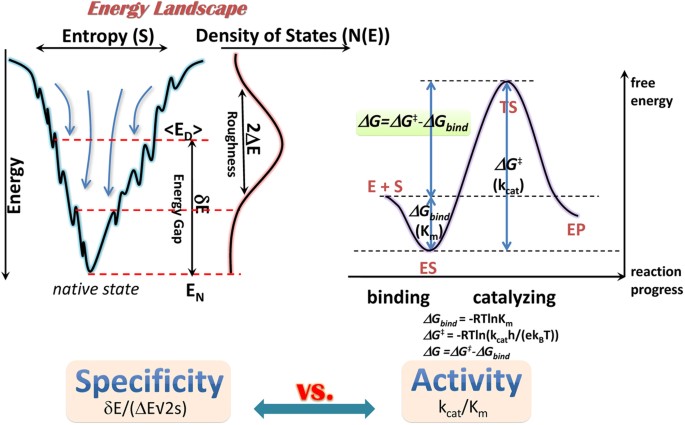
Energy Landscape Topography Reveals the Underlying Link Between Binding Specificity and Activity of Enzymes | Scientific Reports

Minimizing the Entropy Penalty for Ligand Binding: Lessons from the Molecular Recognition of the Histo Blood‐Group Antigens by Human Galectin‐3 - Gimeno - 2019 - Angewandte Chemie International Edition - Wiley Online Library

Water Networks Contribute to Enthalpy/Entropy Compensation in Protein–Ligand Binding | Journal of the American Chemical Society

Entropy–enthalpy transduction caused by conformational shifts can obscure the forces driving protein–ligand binding | PNAS

Binding Energy and Free Energy of Calcium Ion to Calmodulin EF-Hands with the Drude Polarizable Force Field | ACS Physical Chemistry Au

Left: Mean change in total enthalpy upon binding, hDH bind i, of each... | Download Scientific Diagram

Local potential energy: A novel QTAIM tool to quantify the binding energy of classical hydrogen bonds - ScienceDirect