Structural Alert/Reactive Metabolite Concept as Applied in Medicinal Chemistry to Mitigate the Risk of Idiosyncratic Drug Toxicity: A Perspective Based on the Critical Examination of Trends in the Top 200 Drugs Marketed

FDA's Interpretation Of The “Deemed To Be A License” Provision Of The Biologics Price Competition An | Contract Pharma
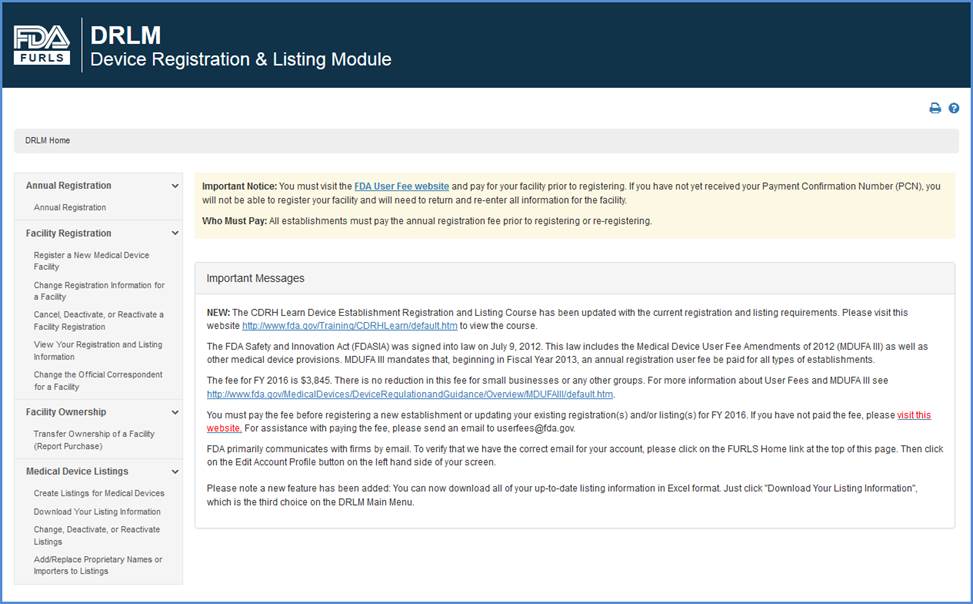
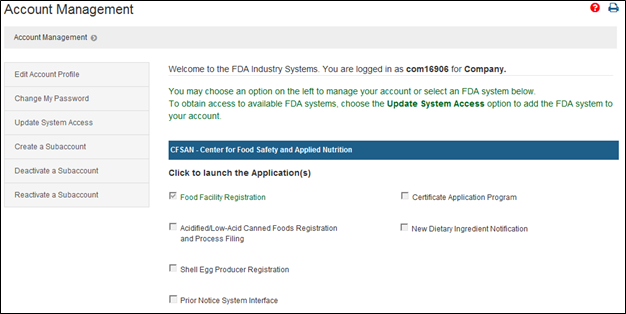
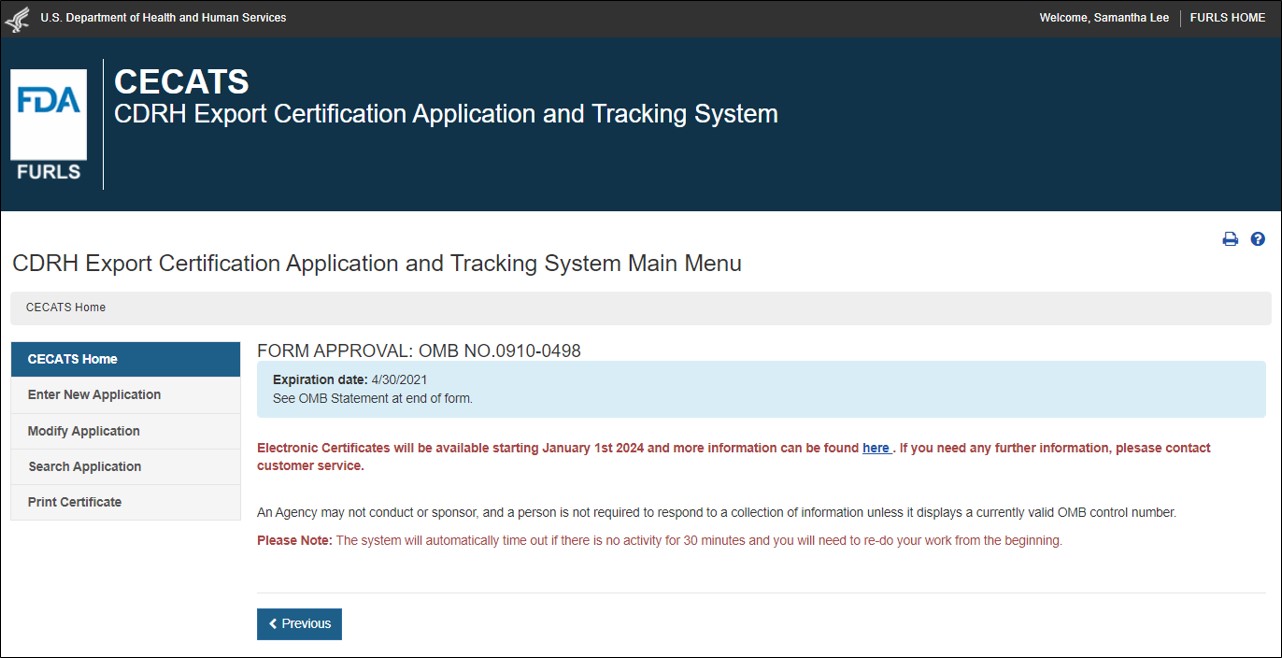
FDA Food Facility Registration is required under laws created by both the Bioterrorism Act of 2003 and Food Safety Modernization
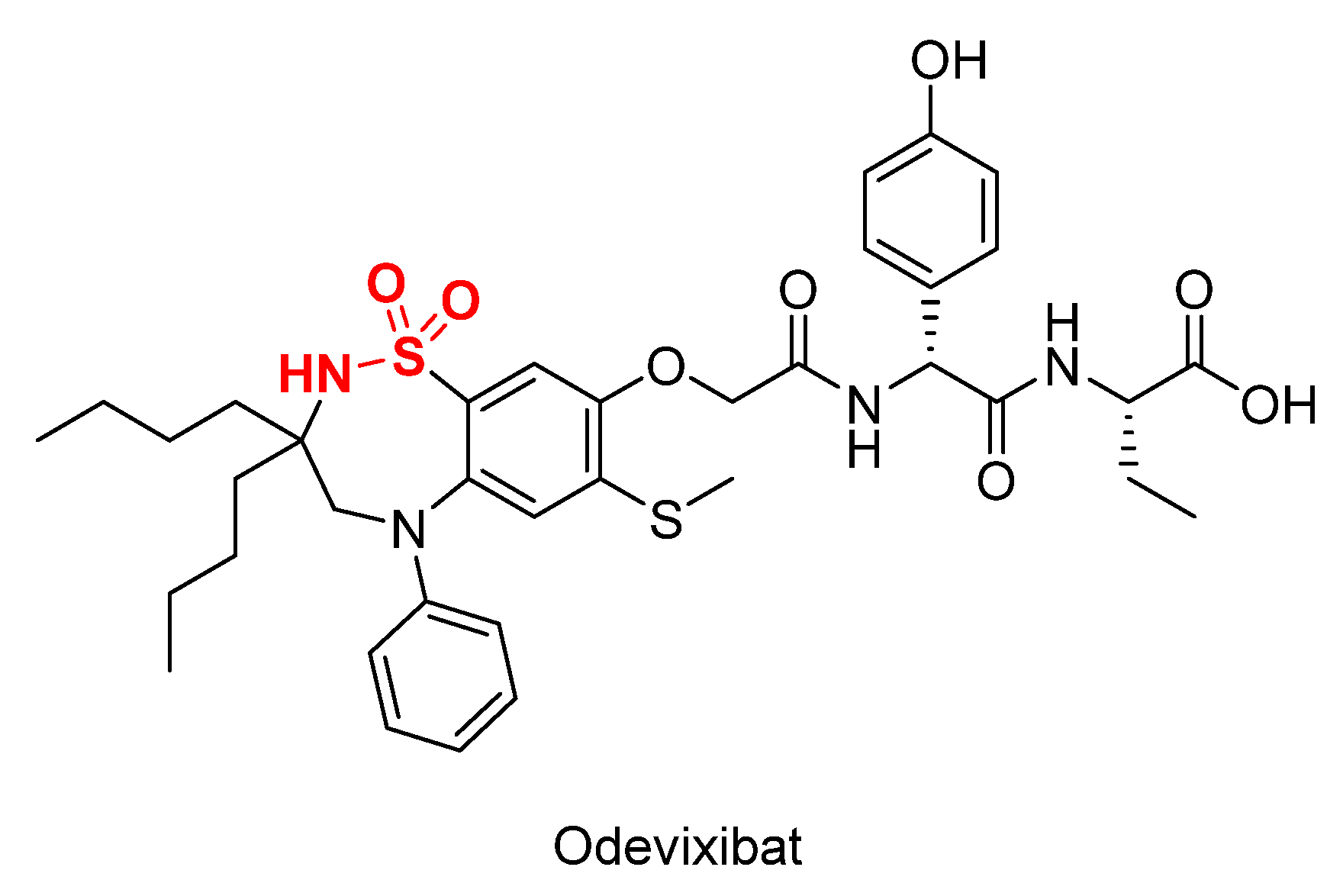
Pharmaceutics | Free Full-Text | FDA-Approved Small Molecules in 2022: Clinical Uses and Their Synthesis
FDA Emergency Use Authorization: A Brief History From 9/11 to COVID-19 - Food and Drug Law Institute (FDLI)

Establishing an FDA office in the Middle East/North Africa: An Abraham Accords Initiative - Food and Drug Law Institute (FDLI)

Meeting FDA Guidance recommendations for replication-competent virus and insertional oncogenesis testing: Molecular Therapy - Methods & Clinical Development